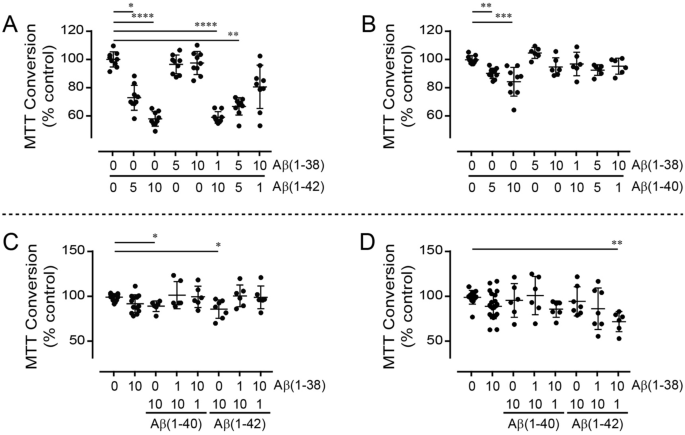
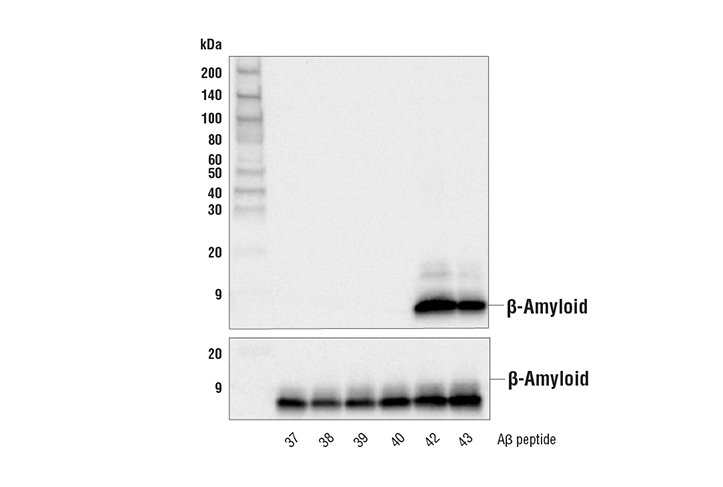
The Aβ(1–38) peptide is a negative regulator of the Aβ(1–42) peptide implicated in Alzheimer disease progression | Scientific Reports

Solution NMR Studies of the Aβ(1−40) and Aβ(1−42) Peptides Establish that the Met35 Oxidation State Affects the Mechanism of Amyloid Formation | Journal of the American Chemical Society

Figure 1 from Amyloid-beta(1-42) rapidly forms protofibrils and oligomers by distinct pathways in low concentrations of sodium dodecylsulfate. | Semantic Scholar
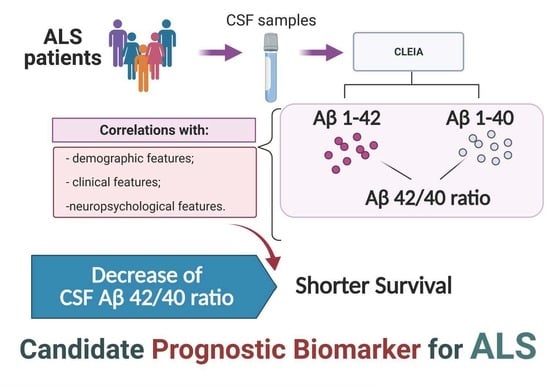
Brain Sciences | Free Full-Text | Prognostic Role of CSF β-amyloid 1–42/1–40 Ratio in Patients Affected by Amyotrophic Lateral Sclerosis

Comparison of Alzheimer Aβ(1–40) and Aβ(1–42) amyloid fibrils reveals similar protofilament structures | PNAS
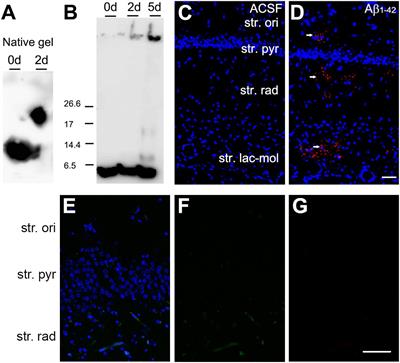
Frontiers | The Acute Effects of Amyloid-Beta1–42 on Glutamatergic Receptor and Transporter Expression in the Mouse Hippocampus

Neurotoxicity and Memory Deficits Induced by Soluble Low-Molecular-Weight Amyloid-β1–42 Oligomers Are Revealed In Vivo by Using a Novel Animal Model | Journal of Neuroscience
Assessments of Aβ-(1-42) under oligomer-and fibril-forming conditions... | Download Scientific Diagram
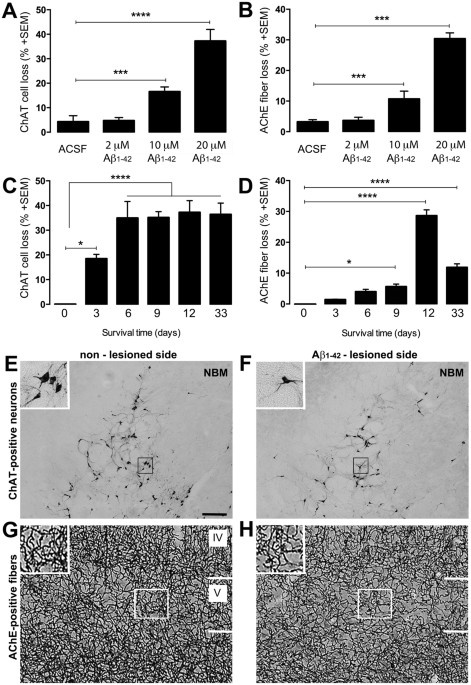
Treatment of beta amyloid 1–42 (Aβ1–42)-induced basal forebrain cholinergic damage by a non-classical estrogen signaling activator in vivo | Scientific Reports

Truncated Amyloid-β(11–40/42) from Alzheimer Disease Binds Cu2+ with a Femtomolar Affinity and Influences Fiber Assembly* - Journal of Biological Chemistry
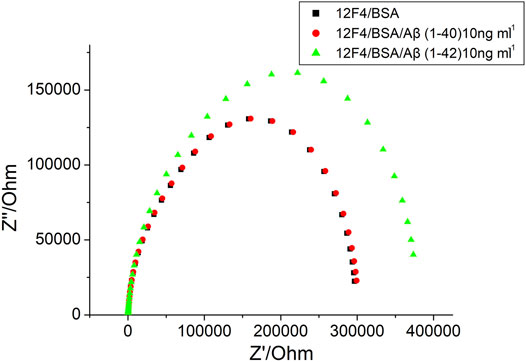
Frontiers | Detection of Amyloid-β(1–42) Aggregation With a Nanostructured Electrochemical Sandwich Immunoassay Biosensor

Figure 1 from Amyloid-beta(1-42) rapidly forms protofibrils and oligomers by distinct pathways in low concentrations of sodium dodecylsulfate. | Semantic Scholar

Mixing Aβ(1–40) and Aβ(1–42) peptides generates unique amyloid fibrils - Chemical Communications (RSC Publishing)


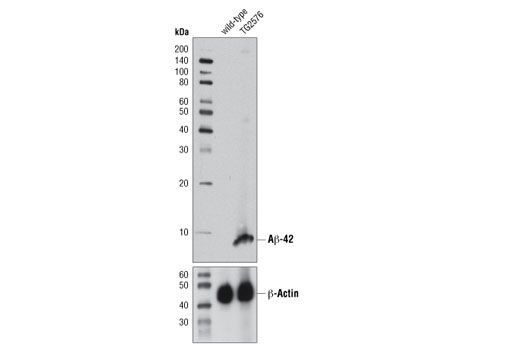

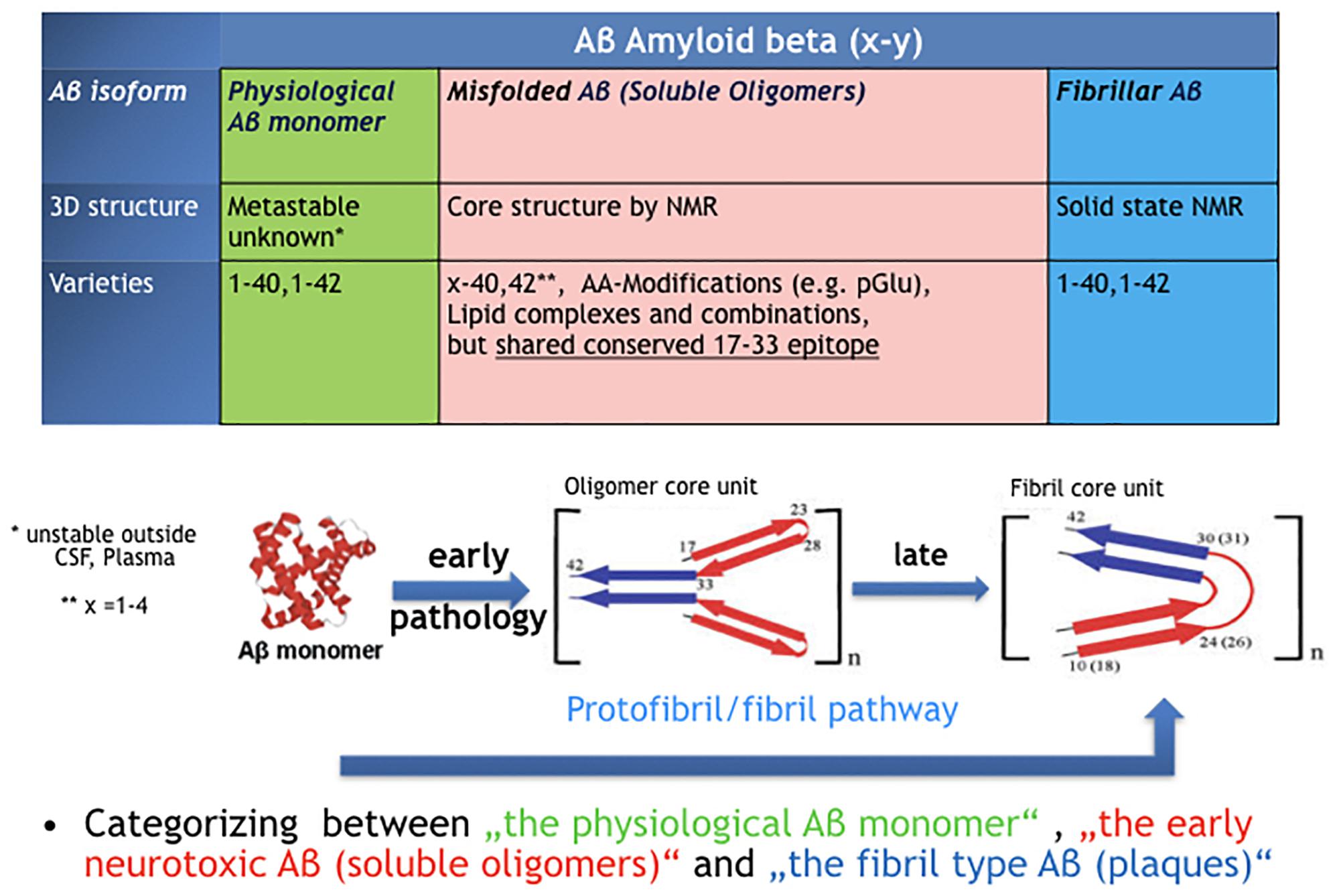




![Amyloid Beta protein[1-42] - an overview | ScienceDirect Topics Amyloid Beta protein[1-42] - an overview | ScienceDirect Topics](https://ars.els-cdn.com/content/image/1-s2.0-S0074774219301035-f05-06-9780128175309.jpg)